S. Abir-Khalil,1 S. Zaîmi,1 M.A. Tazi,2 S. Bendahmane,1 O. Bensaoud1 and M. Benomar1
معدَّل انتشار ارتفاع ضغط الدم لدى رؤية المعطف الأبيض والمنبئات به في قاعدة بيانات ضخمة لرصد ضغط الدم لمرضى العيادات الخارجية
سعدية عبير خليل، سهام زعيمي، محمد عدنان التازي، سندس بندحمان، وفاء بنسعود، محمد بنعمر
الخلاصـة: هدفت هذه الدراسة إلى التعرف على معدَّل انتشار تأثير المعطف الأبيض وارتفاع ضغط الدم لدى رؤيته، وقد اختار الباحثون عدداً من المتغيرات السريرية (الإكلينيكية) بوصفها منبئات بارتفاع ضغط الدم لدى رؤية المعطف الأبيض، وقد أجرى الباحثون مراقبة لضغط الدم شملت 2462 مريضاً خارجياً إما لأن المريض مصاب بارتفاع ضغط الدم الحدّي (المجموعة 1) أو من أجل تقيـيم المعالجة بخافضات ضغط الدم (المجموعة 2) أو لأنه مصاب بارتفاع ضغط الدم (المجموعة 3). ومن بين الذين شملتهم الدراسة ظهر لدى 33% من المرضى ارتفاع في ضغط الدم لدى رؤية المعطف الأبيض و32.8% تبين أن لديهم ارتفاع ضغط الدم الحدّي (المجموعة 1) و37% ممن خضعوا لتقيـيم المعالجة بخافضات ضغط الدم (المجموعة 2). وتبين للباحثين، بإجراء التحليل المتعدد المتغيرات أن الجنس ودرجة ارتفاع ضغط الدم من المنبئات المستقلة عن غيرها لحدوث تأثير المعطف الأبيض لدى المجموعتين 1 و2.
ABSTRACT: The objective of this study was to determine both the prevalence of white-coat effect and white-coat hypertension (WCH) and which selected clinical variables were predictors of WCH. A total of 2462 patients underwent ambulatory blood pressure monitoring either in borderline hypertension (group 1) or for assessment of antihypertensive treatment (group 2) or for hypotension (group 3). In the overall population 33.0% of patients showed WCH, 32.8% in group 1 and 37.0% in group 2. In multivariate analysis, sex and grade of hypertension were independent predictors of WCH in groups 1 and 2.
Prévalence et facteurs prédictifs de l’hypertension « de la blouse blanche » dans une importante base de données de mesure ambulatoire de la pression artérielle
RÉSUMÉ: Cette étude avait pour objectif de déterminer la prévalence de l’effet blouse blanche et de l’hypertension de la blouse blanche, et de recenser les variables cliniques qui sont des facteurs prédictifs de ce type d’hypertension. Au total, 2 462 sujets ont fait l’objet d’une mesure ambulatoire de la pression artérielle, en raison d’une hypertension légère (groupe 1), aux fins de l’évaluation d’un traitement antihypertenseur (groupe 2), ou en raison d’une hypotension (groupe 3). Dans la population générale, 33,0 % des sujets présentaient une hypertension de la blouse blanche, 32,8 % dans le groupe 1 et 37,0 % dans le groupe 2. Dans l’analyse multivariée, le sexe et le niveau d’hypertension étaient des facteurs prédictifs indépendants d’hypertension de la blouse blanche .
1Moroccan League for the Control of Cardiovascular Diseases, Department of Cardiology, Avicenne Hospital, Rabat, Morocco (Correspondence to:
2Directorate of Epidemiology and Control Diseases, Ministry of Health, Rabat, Morocco.
Received: 11/07/06; accepted: 31/10/06
EMHJ, 2009, 15(2): 400-407
Introduction
White-coat effect (WCE) and white-coat hypertension (WCH) are common in medical practice. WCE is defined as the transient rise in blood pressure (BP) from before to during the clinic visit, whereas WCH (also referred to as “office hypertension” or “isolated clinical hypertension”) is generally defined as persistently elevated office BP in the presence of a normal BP outside the office, regardless of the extent of the WCE [1]. Recognition of WCH and WCE is important because a diagnosis of hypertension or resistant hypertension can be misapplied and lifelong drug therapy may be inappropriately prescribed. WCH can itself carry an increased risk of mortality and cardiovascular events [2,3]. It is also frequent in patients with obstructive sleep apnoea–hypopnoea syndrome [4].
Ambulatory blood pressure monitoring (ABPM) has been recognized as a better predictor of risk for organ damage and morbid events than clinic BP monitoring [5–7]. Many patients with high BP in the physician’s office using antihypertensive medication are actually normotensive elsewhere. WCH seems to occur in 24%–39% of the general hypertensive population [8,9]
In Morocco, no study has estimated the prevalence of WCH and WCE or defined their determinants. We aimed to determine the prevalence and characteristics of WCH and WCE in a referral hospital in our country and to assess whether selected clinical variables are predictors of WCH and whether these might identify patients with WCH independently of ABPM.
Methods
Subjects
All patients referred to the cardiology unit at Ibn Sina Teaching Hospital, Rabat, Morocco between January 1999 and March 2001 were considered for ABPM. Patients in whom BP recordings were < 70% of the expected number of readings regarded as valid by the machine software [10] and/or showed no valid readings for ≥ 2 hours were excluded from the analysis. The final study group was 2462 patients for whom ABPM data were accepted for further analysis. Information on age, sex, diabetes, body mass index (BMI) and use of antihypertensive medication at baseline was obtained for every patient.
Measurements
Before beginning ABPM, the patient had a BP measurement performed by the doctor in the blood pressure unit. BP was measured with a mercury sphygmomanometer with appropriate cuff and after 5 minutes sitting quietly. Two measurements were taken and the average of the 2 values (using the higher pressure arm) was taken as the clinic BP. Hypertension was classified into 3 grades, as recommended by the World Health Organization (grade 1 systolic/diastolic BP: 140–159/90–99 mmHg; grade 2 systolic/diastolic BP: 160–179/100–109 mmHg; grade 3 systolic/diastolic BP ≥ 180/≥ 110 mmHg) [11].
The 24-hour ABPM was done on a typical weekday with the SpaceLabs 90207 system; normal daily activities were allowed. BP was recorded every 15 minutes during the day (from 07:00 to 22:00) and every 30 minutes during the night (from 22:00 to 07:00). Mean systolic BP and diastolic BP were calculated for daytime, night-time and for 24 hours. “Non-dippers” were defined by a reduction in systolic BP < 10% from day to night, and the patients falling outside this definition were considered “dippers”.
Using a definition of normal BP outside the medical setting [12,13] and the definition of WCH proposed by Verdecchia et al. [1], patients were classified as having WCH if their clinic systolic or diastolic BP was ≥ 140/90 mmHg and their mean daytime ambulatory systolic and diastolic BP was < 135/85 mmHg. They were considered as having sustained hypertension if their mean daytime ambulatory systolic or diastolic BP was ≥ 135/85 mmHg. The difference between clinic BP and mean daytime ambulatory BP was used to quantify the WCE: patients were considered as havingWCE if this difference was ≥ 5 mmHg.
Statistical analysis
Data are reported as mean and standard deviation (SD). Standard descriptive and comparative statistical analyses are reported. We used the Student t-test to test for significant differences between means for continuous variables and the chi-squared test to test for significant differences for categorical variables. To determine independent clinical predictive factors of WCH, we included in a multivariate logistic regression the clinical variables obtained before ambulatory monitoring that were significantly associated with WCH in univariate analysis. P-value < 0.05 was considered significant
Results
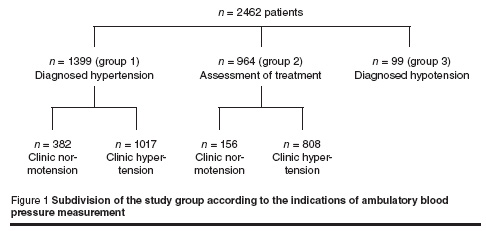
The 2462 patients accepted for ABPM analysis were divided into 3 groups: 1399 were diagnosed with hypertension (group 1), 964 had indications for antihypertensive treatment based on 3 clinic readings of high blood pressure (group 2), and 99 were diagnosed with hypotension with systolic BP < 94 mmHg and/or mean blood pressure < 70 mmHg with symptoms (group 3). Nevertheless, the measure of BP in our unit identified 382 patients in group 1 and 156 patients in group 2 whose clinic BP was normotensive (< 140/90 mmHg without hypotension) (Figure 1). Table 1 shows the main descriptive data in the overall study population.
White-coat effect
The mean difference between clinic and daytime ambulatory BP was 18.2 mmHg (SD 15.3) for systolic pressure and 10.2 mmHg (SD 9.3) for diastolic pressure (Table 1). Using the threshold value of 5 mmHg, the prevalence of WCE was 88.0% in the total group, 88.4% in group 1, 88.9% in group 2 and 66.7% in group 3. Differences between clinic and daytime ambulatory BP were significantly lower in the later group compared with the other groups (P < 0.0001) (mean difference in systolic/diastolic BP was 2/6.4 mmHg in group 3, 17/9.8 mmHg in group 1 and 21/11 mmHg in group 2).
On average, patients with WCE were older (P < 0.0001) than those without WCE and had a higher BMI (P = 0.03) (Table 2). Clinic systolic and diastolic BP were considerably higher in the WCE group, while ambulatory systolic and diastolic BP was significantly lower in this group and the percentage of dippers was higher. There were no significant differences between the groups in terms of sex or the presence of diabetes.
White-coat hypertension
Overall, 817 (33.2%) patients showed WCH, 460 (32.8%) in group 1 and 357 (37.0%) in group 2. WCH was compared with sustained hypertension in group 1 and group 2. Women were more likely to have WCH (Table 3) (P < 0.0001). The intensity of WCE was significantly higher, whereas clinic systolic and diastolic BP as well as ambulatory systolic and diastolic BP were considerably lower, in the WCH group (systolic/diastolic 24.0/15.1 mmHg versus 17.8/8.7 mmHg in group 1 and systolic/diastolic 28.4/16.0 mmHg versus 19.6 9.2 mmHg in group 2). The proportion of non-dippers was significantly higher in the WCH group. In addition, WCH patients showed significantly elevated BMI compared with patients with sustained hypertension. No significant differences were found in terms of age and presence of diabetes.
In order to determine factors predictive of WCH in groups 1 and 2, separate multivariate analysis for both groups was done using the clinical variables obtained before ABPM which were significantly associated with WCH in univariate analysis (sex, BMI level and clinic BP level). Sex and clinic BP level were significant predictors of WCH in both groups; women and those with hypertension grade 1 (systolic BP 140–159 mmHg or diastolic BP 90–99 mmHg) were more likely to exhibit WCH (Table 4 and Table 5). Among patients with clinic hypertension, 75.0% of women with grade 1 hypertension had WCH versus 56.0% of men. There were 22.8% of men and 31.2% of women with more serious grades of hypertension (grade 2 or 3) who showed WCH response.
Discussion
The WCE and WCH differ in their definitions, physiological mechanisms and clinical significance [14]. WCE is a phenomenon which may be expected to occur in the majority of patients, even among hypotensive subjects [15]. In our sample, using a threshold of 5 mmHg, the prevalence of WCE was 88.0% in the overall population and 66.7% in the hypotensive group. Mancia et al. have shown that the rise in intra-arterial BP during the clinic visit is 27/14 mmHg on average [15]. In the current study, the magnitude of the WCE was lower on average (18/10 mmHg), presumably because we used non-invasive ABPM.
Depending on the upper limit for mean daytime ambulatory BP, the prevalence of WCH can range from 18% to 60% [16,17]. Using the definitions of normal BP outside the medical setting and of WCH proposed at the last international consensus conference on ambulatory BP [1,12,13]; 33% of our patients were found to have WCH.
Few studies have been conducted on patients receiving antihypertensive treatment [18–20]. In the present study, 37% of such patients had WCH. This phenomenon remains a consideration even among patients who are poorly controlled with antihypertensive treatment. We showed that the intensity of WCE is greater, on average, in subjects with WCH than it is in those with sustained hypertension (systolic/diastolic 24.0/15.1 mmHg versus 17.8/8.7 mmHg in group 1 and systolic/diastolic 28.4/16.0 mmHg versus 19.6 /9.2 mmHg in group 2), as shown by others [21].
In our study as well as in the literature, a large difference between clinic and ambulatory BP was observed in older patients [22,23] and was associated with high values of clinic BP and low values of ambulatory BP [21,24]. This relation provides an explanation for the prognostic significance of this difference which cannot predict cardiovascular morbidity and mortality in subjects with essential hypertension [22]. Several investigators have attempted to identify the clinical, psychological and demographic predictors of WCH, but findings have been conflicting or not significant [25]. Of the variables examined in the present study, only female sex and mild hypertension were predictive of WCH. Mild hypertension was a predictor factor of WCH in 1 study [20]; however it is known that the prevalence of WCH decreases with increasing Joint National Committee V stage of hypertension [21]. Indeed, although the intensity of WCE is higher in patients with WCH [21], the WCE does not necessarily show a direct association with WCH [22]. In other words, a small WCE may lead to WCH in subjects with mild hypertension.
Sex is an established independent predictor of WCH [19,20]. In our sample, women were approximately twice as likely as men to exhibit WCH; the reason for the sex difference is unclear and should be investigated in future studies. However, it is important to emphasize that WCH cannot be predicted reliably; clinical variables are not enough to substitute (as diagnostic tools) for ABPM, which must be seen as a useful instrument in clinical practice [26].
Several studies have suggested that WCH is benign and that antihypertensive therapy is not required [27] whereas others have suggested that it may not be innocent [1,2,28]. Non-dipper status was significantly predominant in our WCH group; this supports the hypothesis that WCH is not an innocent condition. So, we suggest that patients with WCH require regular and accurate follow-up; remonitoring may be helpful in this condition [29]. Large cohort studies are needed to establish whether the long-term risk of cardiovascular disease is comparable between subjects with WCH and clinically normotensive individuals.
References
- Verdecchia P et al. Properly defining white coat hypertension. European heart journal, 2002, 23:106–9.
- Mancia G et al. Long-term risk of mortality associated with selective and combined elevation in office, home, and ambulatory blood pressure. Hypertension, 2006, 47:846–53.
- Verdecchia P et al. Short- and long-term incidence of stroke in white-coat hypertension. Hypertension, 2005, 45:203–8.
- Garcia-Rio F et al. White coat hypertension in patients with obstructive sleep apnea-hypopnea syndrome. Chest, 2004, 125(3):817–2.
- Martinez M et al. Frequency and determinants of white coat hypertension in mild to moderate hypertension: a primary care-based study. American journal hypertension, 1999, 12:251–9.
- Tochikubo O et al. Estimation of base blood pressure by using a new device in the outpatient clinic. Hypertension research, 2006, 29(4):233–41.
- Helvaci MR, Seyhanli M. What a high prevalence of white coat hypertension in society! Internal medicine, 2006, 45(10):671–4.
- Kuriyama S et al. Morning blood pressure predicts hypertensive organ damage in patients with renal diseases: effect of intensive antihypertensive therapy in patients with diabetic nephropathy. Internal medicine, 2005, 44(12):1239–46.
- Torun D et al. The frequency of combined target organ damage and the beneficial effect of ambulatory blood pressure monitoring in never treated mild-to-moderate hypertensive patients. International heart journal, 2005, 46(6):1073–82.
- Casadei R et al. 24-hour blood pressure monitoring: evaluation of Spacelabs 5300 monitor by comparison with intra-arterial blood pressure recording in ambulant subjects. Journal of hypertension, 1988, 6:797–803.
- 1999 World Health Organization–International Society of Hypertension guidelines for the management of hypertension. Guidelines subcommittee. Journal of hypertension, 1999, 17:151–83.
- Staessen JA et al. Predicting cardiovascular risk using conventional vs ambulatory blood pressure in older patients with systolic hypertension. Systolic hypertension in Europe Trial Investigators. Journal of the American Medical Association, 1999, 282:539–46.
- Pickering TG et al. Blood pressure monitoring. Task force V: White-coat hypertesion. Blood pressure monitoring, 1999, 4:333–41.
- Verdecchia P et al. Prognostic significance of the white-coat effect. Hypertension, 1997, 29:1218–24.
- Mancia C et al. Effect of blood pressure measured by the doctor on patient’s blood pressure and heart rate. Lancet, 1983, 2:695–8.
- Verdecchia P et al. Variability between current definitions of normal ambulatory blood pressure: implications in the assessment of white-coat hypertension. Hypertension, 1992, 20:555–62.
- Pierdomenico SD et al. White-coat hypertension in patients with newly diagnosed hypertension: evaluation of prevalence by ambulatory monitoring and impact on cost of health care. European heart journal, 1995, 16:692–7.
- Myers MG, Reeves RA. White-coat phenomenon in patients receiving antihypertensive therapy. American journal of hypertension, 1991, 4:844–9.
- Myers MG, Reeves RA. White-coat effect in treated hypertensive patients: sex differences. Journal of human hypertension, 1995, 9:729–33.
- MacDonald MB et al. Prevalence and predictors of white-coat response in patients with treated hypertension. Canadian Medical Association journal
- Verdecchia P et al. White-coat hypertension and white-coat effect: similarities and differences. American journal of hypertension, 1995, 8:790–8.
- Verdecchia P et al. Prognostic significance of the white-coat effect. Hypertension
- Mansoor GA, McCabe EJ, White WB. Determinants of the white-coat effect in hypertensive subjects. Journal of human hypertension, 1996, 10:87–92.
- Pickering TG. Blood pressure measurement and detection of hypertension. Lancet, 1994, 344:31–5.
- Siegel WC, Blumenthal JA, Divine GW. Physiological, psychological, and behavioral factors and white-coat hypertension. Hypertension, 1990, 16:140–6.
- Staessen JA et al. The Ambulatory Blood Pressure Monitoring and Treatment of Hypertension Investigators. Antihypertensive treatment based on conventional or ambulatory blood pressure measurement. Journal of the American Medical Association, 1999, 78:1065–72.
- Khattar RS, Senior R, Lahiri A. cardiovascular outcome in white-coat versus sustained mild hypertension: a 10 year follow-up study. Circulation, 1998, 98:1892–7.
- Bidlingmeyer I et al. Isolated office hypertension: a prehypertensive state? Journal of hypertensionn
- O’Brien ET. Blood pressure measurement. British medical journall, 2001, 322:1110–4.








