M.S. Alkhalifa,1 S.A. Ibrahim2 and S.H. Osman3
ABSTRACT We determined the pattern and severity of valvular involvement in 100 Sudanese patients with rheumatic heart disease (RHD) in 2 centres in Khartoum in 2003. All the patients underwent history taking, physical examination and Doppler echocardiography. The commonest lesions were mitral regurgitation (84%) and aortic regurgitation (40%). Mitral stenosis alone was uncommon (9%) which could be due to a low detection rate related to the slow stenotic process and the subtle early signs. This, together with the inconsistent history of rheumatic fever, may lead to an underestimate of the prevalence of RHD. There was a strong correlation between severity of the lesion and irregular prophylaxis (P < 0.001).
Caractéristiques et gravité des lésions valvulaires rhumatismales chez des enfants à Khartoum au Soudan
RÉSUMÉ Nous avons déterminé les caractéristiques et la gravité de l’atteinte valvulaire chez 100 patients soudanais souffrant de cardiopathie rhumatismale dans deux centres de Khartoum en 2003. Tous les patients ont fait l’objet d’un interrogatoire médical, d’un examen physique et d’une échocardiographie Doppler. Les lésions les plus courantes étaient la régurgitation mitrale (84 %) et la régurgitation aortique (40 %). La sténose mitrale seule était rare (9 %), ce qui pouvait être dû à un faible taux de détection lié à la lenteur du processus sténotique et au caractère imperceptible des premiers signes ; ces éléments, combinés à l’incertitude sur les antécédents de rhumatisme articulaire aigu, peuvent mener à une sous-estimation de la prévalence de la cardiopathie rhumatismale. Il existait une forte corrélation entre la gravité de la lésion et l’irrégularité de la prophylaxie (p < 0,001).
1Omdurman Islamic University, Elshaab Teaching Hosptial, Khartoum, Sudan (Correspondence to M.S. Alkhalifa:
2Department of Paediatrics, University of Khartoum, Khartoum, Sudan 3Cardiology Unit, Gafar Ibn Oaf Children’s Hospital, Khartoum, Sudan.
Received: 19/03/06; accepted: 14/06/06
EMHJ, 2008, 14(5): 1015-1021
Introduction
Acute and chronic rheumatic heart disease is the commonest cause of acquired heart disease in children. While this disease has almost disappeared in developed countries, it is still a leading cause of morbidity and mortality from heart disease in developing countries [1–3].
Recent reports from developing countries have documented the incidence of rheumatic fever as high as 206/100 000 population and rheumatic heart disease (RHD) prevalence as high as 18.6/1000 population, although there are variations between countries and different areas within the same country [4–12].
The clinical picture, morbidity and mortality, frequency and speed of development of an established valve disease differ geographically and are affected primarily by the socioeconomic and medical background of the patients [13]. In developing countries the course is more violent with severe pathological valvular lesions necessitating surgery in young children [14].
In chronic RHD echocardiography is the fundamental tool to determine the valves involved and the pattern of involvement, and to asses the severity of the lesion and to rule out non-rheumatic causes [15–17].
A study of the pattern of RHD in 100 Sudanese children using clinical examination, ECG and chest X-ray showed that the mitral valve was the commonest affected valve followed by mitral valve + aortic valve. The major complication was heart failure, which was mainly treated with frusemide and digoxin [18]. There have been no previous studies of RHD in Sudan using echocardiography as a diagnostic tool. The objective of this study was to determine the pattern and severity of valvular involvement in children with RHD using echocardiography.
Methods
We recruited 100 patients with documented RHD up to the age of 16 years who were referred to the outpatient clinic at Ahmed Gasim Cardiac Centre and Al Shaab Teaching Hospital during the period between June and November 2003 in this descriptive, cross-sectional hospital-based study.
A thorough history was taken which included personal data, previous history of rheumatic fever (according to the Jones criteria), duration and regularity in prophylaxis with long-acting penicillin over the last 12 months, presenting complaints and other information such as housing conditions. Physical examination was done including anthropometric measurement and cardiovascular system examination.
Echo Doppler evaluation for cardiac lesions was performed; the valves involved, pattern of valvular involvement and severity of the lesions were assessed.
The following echo criteria for severity were chosen based on the criteria used in our laboratory:
- Increase of the left ventricular end diastolic dimension was considered severe if ≥ 6.5 cm.
- Left ventricular ejection fraction was considered reduced if < 60%.
- Left atrial enlargement was considered severe if ≥ 6 cm.
- Mitral stenosis was considered severe if the mitral valve area was < 1 cm.
- Severe pulmonary hypertension was defined as tricuspid regurgitation gradient > 60 mmHg.
Statistical analysis
The data were analysed using SPSS, version 2. Significance testing of difference between proportions was done using the chisquared test at the 95% confidence level.
Results
There were slightly more females (55%) than males, the majority of patients were in the age range 13–16 years (69%) (minimum 5 and maximum 16 years) and were from periurban and rural areas (93%). The majority of patients (94%) came from a low socioeconomic background and were from rural areas or lived on the outskirts of cities Overcrowded living conditions with more than 6 persons per room were reported in 63% of the cases.
Isolated mitral valve involvement was found in 60% of the patients, while isolated aortic valve disease was seen in only 7%.
The commonest lesions were mitral regurgitation and aortic regurgitation. This was followed by mitral stenosis, while aortic stenosis was very rare and was not detected as an isolated lesion. The commonest isolated lesion was mitral regurgitation (Table 1).
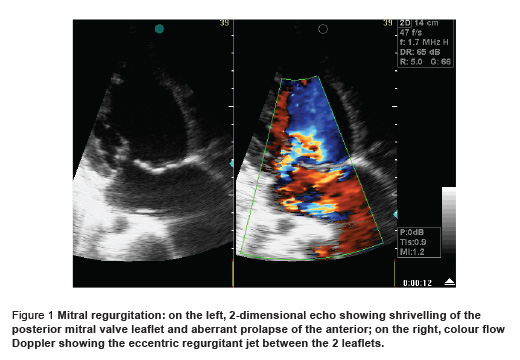
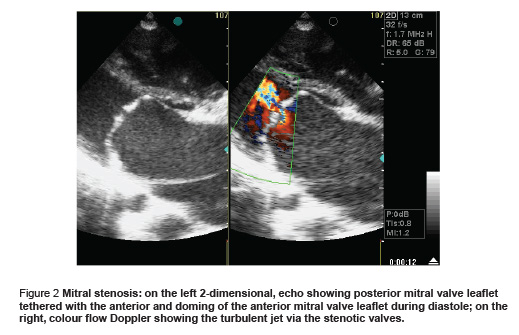
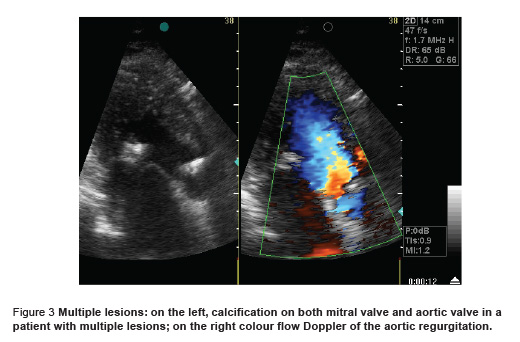
The morphology of the valve and the characteristics of the lesion as seen by echocardiography were generally fixed for the type of lesion. Most patients with mitral regurgitation showed thickening and shrivelling of the posterior mitral valve leaflet and shortening of the chordate with aberrant prolapse of the anterior mitral valve leaflet and an eccentric regurgitant jet towards the left atrial lateral wall (Figure 1). Most patients with mitral stenosis showed thickening of both leaflets with pliable body and commissural fusion with the posterior mitral valve leaflet tethered with the anterior and moving forwards with it with doming of the anterior mitral valve leaflet during diastole (Figure 2). Most patients with combined mitral stenosis and mitral regurgitation showed mixed features of both patterns with some calcification. Aortic regurgitation showed thickening and retraction of the cusps. Multiple lesions showed mixed pattern with deformed valves with some calcification (Figure 3).
Regarding severity, 51% of the patients had such severe lesions as to require intervention. This included all the patients with combined mitral regurgitation + aortic regurgitation and all the patients with multiple lesions. In addition, all the patients with mitral stenosis alone had severe lesions, while 9 (25%) patients with mitral regurgitation alone had severe lesions (Table 2).
The severity also correlated with a lack of compliance with secondary prophylaxis. Of the 18 patients with severe mitral valve disease, 61% had not complied with secondary prophylaxis compared with 39% who had complied (P < 0.001). There was no correlation of severity with history of rheumatic fever; 33.3% of the 18 patients with severe mitral valve disease had a history of rheumatic fever, while 66.7% did not (P = 0.202).
Discussion
The study showed that RHD is still a disease of the low socioeconomic classes with 94% of patients of low socioeconomic status. This agrees with the findings from India, Egypt and Hawaii [6–9,11,9,19].
The mitral valve was the commonest valve involved in our patients, followed by combined mitral valve and aortic valve involvement. Isolated aortic valve disease was rare which is similar to the pattern of cases described in Ethiopia, Nigeria and India [20–22].
The type of valvular involvement differed from that in adults as the majority of our patients had regurgitant lesions, while isolated mitral stenosis, which is the commonest lesion in adults, was detected in only 9% of our patients. This could be explained by the very slow stenotic process and the subtle signs of early mitral stenosis, which may be easily missed.
History of rheumatic fever was not consistent in our patients as 66.7% did not have such a history; the same results were obtained from Egypt and India [9,23]. This, together with the low detection rate of mitral stenosis, may lead to an underestimation of the prevalence of RHD in children if screened by clinical features alone. This suggests that the use of echocardiography may be warranted as a screening tool for early detection of cases of RHD, at least for children who have a history of recurrent tonsillitis or family history of rheumatic fever or RHD.
The pattern of valvular involvement is particularly important for mitral stenosis and fortunately in this study the pattern showed features favourable for percutaneous transmittal commissurotomy. For other valvular lesions the pattern is not so important as the current option is valve replacement if the lesion becomes severe, irrespective of the pattern. The factors that predispose to a specific pattern were not studied in our patients but may be immunological or genetic or related to the organism subtypes; this is an area that needs further study.
The severity of the valvular lesions in this study was strongly associated with irregularity in secondary prophylaxis. This might be attributed to an insidious onset or subclinical attacks of rheumatic fever and may reflect a lack of health education. Similar results were obtained from Egypt [9,24]. The severity also correlated with the haemodynamic effect of the lesion and was seen mainly with lesions leading to severe volume overload (combined mitral regurgitation + aortic regurgitation) and in multiple lesions, because patients with multiple lesions had combined mitral regurgitation + aortic regurgitation in addition to mitral stenosis; despite the presence of mitral stenosis, the degree of volume overload is still severe. On the other hand, the severity was less with an isolated regurgitant lesion, e.g. mitral regurgitation, and indeed this was true when the isolated regurgitant lesion was combined with mitral stenosis.
Mitral stenosis alone when detected in our patients was severe enough to mandate intervention and this was seen in the older children (7 of the 9 patients with mitral stenosis were 16 years of age and 2 were 15 years of age). This again may be explained by the early onset but slow and undetected rheumatic process, i.e. missed cases (with no follow-up or secondary prophylaxis) who are detected late when the lesion has become severe and the patient symptomatic.
Recommendations
The following measures need to be carried out together in order to overcome the impact of rheumatic fever and RHD.
- Use of echocardiography as a screening tool for early detection of subtle cases of RHD
- Comprehensive application of the control programme in periurban and rural areas with special emphasis on the regular use of secondary prophylaxis
- Control of volume overload
- Improvement of housing conditions.
Further studies are needed to detect the factors governing the pattern of valvular involvement.
References
- Schwankhaus JD. Preventing rheumatic heart disease in developing countries. Annals of internal medicine, 1994, 121:77.
- Brennan RE, Patel MS. Acute rheumatic fever and rheumatic heart disease in a rural central Australian aboriginal community. Medical journal of Australia, 1990, 153(6):335–8.
- Boon NA, Bloomfield P. The medical management of valvar heart disease. Heart, 2002, 87:395–400.
- Secular IP. Acute and chronic rheumatic heart disease. In: Anderson RH et al., eds. Paediatric cardiology, 1st ed. London, Butter & Tanner Ltd, 1987:1179–215.
- Bernstein D. Rheumatic heart disease. In: Behrman RE, Kliegman RM, Jenson HB, eds. Nelson’s textbook of paediatrics, 16th ed. Philadelphia, WB Saunders Company, 2000:1428–30.
- Gabriel KM, ed. Cardiac and pulmonary management, 1st ed. Pennsylvania, Lea & Febiger, 1993:349–87.
- Cunningham MW. Pathogenesis of group A streptococcal infection. Journal of clinical microbiology, 2000, 13:470–511.
- Eisenberg MJ. Rheumatic heart disease in the developing world: prevalence, prevention and control. European heart journal, 1993, 14(1):122–8.
- Padmavati S. Rheumatic fever and rheumatic heart disease in India at the turn of the century. Indian heart journal, 2001, 53(1):35–7.
- Kumar R et al. A community based rheumatic fever/rheumatic heart disease cohort: twelve-year experience. Indian heart journal, 2002, 54:54–8.
- Jose VJ, Gomathi M. Declining prevalence of rheumatic heart disease in rural school children in India: 2001–2002. Indian heart journal, 2003, 55:158–60.
- Bassili A et al. Profile of secondary prophylaxis among children with rheumatic heart disease in Alexandria, Egypt. Eastern Mediterranean health journal, 2000, 6(2– 3):437–46.
- Hasab A, Jaffer A, Abdulla MR. Rheumatic heart disease among Omani school children. Eastern Mediterranean health journal, 1997, 3(1):17–23.
- Padmavati S. Rheumatic heart disease: prevalence and preventive measures in the Indian subcontinent. Heart, 2001, 86:127–31.
- Al-Munibari AN et al. Prevalence of rheumatic fever and rheumatic heart disease in Yemen. Asian cardiovascular & thoracic annals, 2001, 9:41–4.
- McLaren MJ, Markowitz M, Gerber MA. Rheumatic heart disease in developing countries: the consequence of inadequate prevention. Annals of internal medicine, 1994, 120:243–5.
- Mishra TK et al. Has the prevalence of rheumatic fever/rheumatic heart disease really changed? A hospital-based study. Indian heart journal, 2003, 55(2):152–7.
- Saeed A. The pattern of rheumatic heart disease in children presenting to Khartoum teaching hospital [Thesis]. Khartoum, University of Khartoum, 1983:23–40.
- Steer AC et al. Rheumatic heart disease in school children in Samoa. Archives of disease in childhood, 1999, 81:372.
- Ephrem D, Abegaz B, Muhe L. Profile of cardiac disease in Ethiopian children. East African medical journal, 1990, 67(2):113– 20.
- Onwuchekwa AC, Ugwu EC. Pattern of rheumatic heart disease in adults in Maiduguri—north east Nigeria. Tropical doctor, 1996, 26(2):67–9.
- Vasan RS et al. Echocardiographic evaluation of patients with acute rheumatic fever and rheumatic carditis. Circulation, 1996, 94:73–82.
- Kumar A, Shinha, M. and Shinha DN. Chronic rheumatic heart diseases in Ranchi. Angiology, 1982, 33:141–5.
- WHO programme for the prevention of rheumatic fever/rheumatic heart disease in 16 developing countries: report from Phase I (1986–90). WHO Cardiovascular Diseases Unit and principal investigators. Bulletin of the World Health Organization, 1992, 70(2):213–8.








