Resaerch article
M. Usman,1 M. Moinuddin 1 and R. Ghani 2
الوراثيات الجزيئية لمتلازمة البيتا-ثالاسيمية في باكستان
محمد عثمان، معين الدين، روبينه غني
الخلاصة: أجريت هذه الدراسة الوراثية الجزيئية في مدينة كراتشي بباكستان، في ما بين عامَيْ 2004 و2006 لتقديم دلائل إرشادية لبرامج التشخيص في الفتـرة السابقة للولادة في باكستان. وجُمعت عينات الدم من مرضى مصابين بالبيتا-ثالاسيمية الصغرى (عددهم=200) والبيتا-ثالاسيمية الكبرى (عددهم=150)، وذلك في المستشفيات، ومراكز نقل الدم، والمختبرات من مختلف مقاطعات كراتشي، وهي تمثـِّل خمس مجموعات عرقية كبرى. وأظهر التحليل الجزيئي إحدى عشْرةَ طفرة جينية في جين البيتا-ثالاسيمية، شكلت خمس طفرات منها %88 من إجمالي جينات البيتا-ثالاسيمية التي تم التعرف عليها [وهي IVS-1-5(G-C)، Fr8/9(+G)، Fr41/42(-TTCT)، IVS-1-1(G-T)، وDel619]. ومن الطفرات الأخرى التي تم التعرف عليها: CAP+1، IVS-II-1(G-A)، Cd 5(-CT)، Cd 15(G-A)، Cd 16 وCd 30.
ABSTRACT This molecular genetics study was conducted in Karachi, Pakistan from 2004 to 2006 to provide guidelines for prenatal diagnosis programmes in the country. Blood samples of patients with beta-thalassaemia minor (n = 200) and beta-thalassaemia major (n = 150) were collected from hospitals, transfusion centres and diagnostic laboratories from different districts of Karachi, representing 5 major ethnic groups. Molecular analysis revealed 11 genetic mutations of the beta-thalassaemia gene, among which 5 mutations accounted for 88% of the total beta-thalassaemia genes identified [IVS-1-5 (G–C), Fr 8/9 (+G), Fr 41/42 (–TTCT), IVS-1-1 (G–T) and Del 619]. Other mutations identified were: CAP+1, IVS-II-1 (G–A), Cd 5 (–CT), Cd 15 (G–A), Cd 16 and Cd 30.
Génétique moléculaire de la bêta-thalassémie au Pakistan
RÉSUMÉ Cette étude de génétique moléculaire a été réalisée à Karachi, au Pakistan, entre 2004 et 2006, dans le but de formuler des recommandations applicables aux programmes de diagnostic prénatal du pays. Les prélèvements sanguins de patients atteints de bêta-thalassémie mineure (n = 200) et de bêta-thalassémie majeure (n = 150) ont été recueillis dans des hôpitaux, des centres de transfusion et des laboratoires de diagnostic de différents districts de Karachi, représentant ainsi cinq grands groupes ethniques. L’analyse moléculaire a révélé 11 mutations du gène responsable de la bêta-thalassémie, dont cinq concernaient 88 % des gènes responsables de la bêta-thalassémie identifiés [IVS-1-5 (G – C), Fr 8/9 (+ G), Fr 41/42 (–TTCT), IVS-1-1 (G – T) et Del 619]. Les autres mutations observées ont été les suivantes : CAP + 1, IVS-II-1 (G – A), Cd 5 (–CT), Cd 15 (G – A), Cd 16 et Cd 30.
1Institute of Haematology, Baqai Medical University, Karachi, Pakistan.
2Department of Biochemistry, Baqai Medical University, Karachi, Pakistan (Correspondence to M. Usman:
Received: 25/01/09; accepted: 09/03/09
EMHJ, 2010, 16(9): 972-976
Introduction
Thalassaemia is the most common genetic disorder across the world [1–4]. Thalassaemia was not recognized as a clinical entity until 1925, when Cooley and Lee described a syndrome occurring early in life that was associated with splenomegaly and bone deformities [4,5].
Thalassaemia occurs with a high frequency in a broad belt extending from the Mediterranean basin through to the Middle East, Indian subcontinent and South-East Asia [6–9]. About 3% of the world population (150 million people) are carries of the beta-thalassaemia gene [10–12]. Beta-thalassaemia is also the most common genetic disorder in Pakistan, a country with a population of around 160 million people. The annual rate of population growth is 3% and almost 40% of the population is below 15 years of age [13,14]. There are 5 major ethnic groups: Sindhi, Urdu speaking, Punjabi, Baluchi and Pathan. The Urdu-speaking group also includes 2 large communities, the Memon and the Gujarati. The carrier frequency of beta-thalassaemia is estimated to be around 6% in Pakistani population [15,16].
This molecular genetic study of beta-thalassaemia syndrome in 5 major ethnic groups of Pakistan was established to provide guidelines for prenatal diagnosis programmes in the country.
Methods
Sample
The study group was a convenience sample of 150 patients with beta-thalassaemia major and 200 with beta-thalassaemia minor. The beta-thalassaemia major cases were collected mostly from transfusion centres; these patients had transfusion-dependent thalassaemia and were registered at different transfusion centres of Karachi. Patients had been diagnosed on the basis of complete blood picture, haemoglobin electrophoresis, peripheral blood morphology and clinical signs and symptoms at the time of registration. The beta-thalassaemia minor cases had been identified during screening for beta-thalassaemia minor in the general population and included all major ethnic groups and thalassaemia families (i.e. those with children with transfusion-dependent beta-thalassaemia). These samples were collected from different transfusion centres, hospitals and diagnostic laboratories of Karachi.
Patients were selected to represent the different ethnic groups of Pakistan. The ethnic breakdown was as follows: 80 Punjabi, 50 Pathan, 70 Sindhi, 50 Baluchi and 100 Urdu speaking (Table 1).
Laboratory methods
Venous blood samples were collected into EDTA tubes. All samples were tested by a modified amplification of refractory mutation system (ARMS) [17,18] for the 11 mutations previously reported in the population of the Indian sub-continent. The 11 mutated primer sequences which were used during this study were: IVSI-1 (G–T), IVSI-5 (G–C), IVSII-1 (G–A), Del 619, Fr 41–42 (–TTCT), Fr 8–9 (+G), Fr 16 (–C), Cd 5 (–CT), Cd 30 (G–C), Cd 15 (G–A) and Cap +1 (A–C). DNA was extracted from whole blood by using the Genomic DNA Purification Kit (Gentra Systems, Minneapolis, USA). ARMS primers were designed for detection of normal and mutant DNA. A control pair of primers was included in each assay. Control primers A, B and C were amplified at 861 bp fragments from the 3′ end of the β-globin gene.
Polymerase chain reaction (PCR) was conducted by a modified method in a mixture of 10 mmol/L tris (pH 8.3), 50 mmol/L KCl, 1.5 mmol/L MgCl2. 500 µM of each dNTP, 0.2 µmol/L of each primer, 0.5 units of Taq polymerase and 0.5 to 1 pg of genomic DNA was added to the PCR mixture in a total volume of 20 µL. The modified cycling reaction (DNA Thermal Cycler, Perkin-Elmer/Cetus) was programmed at 94 °C for 1 min. (denature), 65 °C for 1 min. (anneal) and 72 °C for 1.5 min. (extend). After 25 cycles, the samples were incubated for an additional 3 min. at 66 °C [17,19].
PCR products were removed and mixed with 3 μL of a loading buffer and then loaded on 2% agarose gel. The gel was set at 100 volts for 1 hour and then stained with ethidium bromide. After staining, the bands became visible under ultraviolet light. The different mutations were characterized with a 100 bp DNA ladder.
Results
Molecular analysis was carried out for 200 patients with beta-thalassaemia minor and 150 with beta-thalassaemia major. A total of 500 beta-thalassaemia genes were identified. The ethnic breakdown was as follows: 120 genes from Punjabi patients, 70 from Pathans, 90 from Sindhi, 70 from Baluchi and 150 from Urdu-speaking patients (Table 1).
Figure 1 and Figure 2 show sample DNA sequences. The most common mutations were IVS-1-5 (G–C), Fr 8/9 (+G), Fr 41/42 (–TTCT), IVS-1-1 (G–T) and Del 619 and together these comprised 88% of the total beta-thalassaemia genes identified (Table 2).

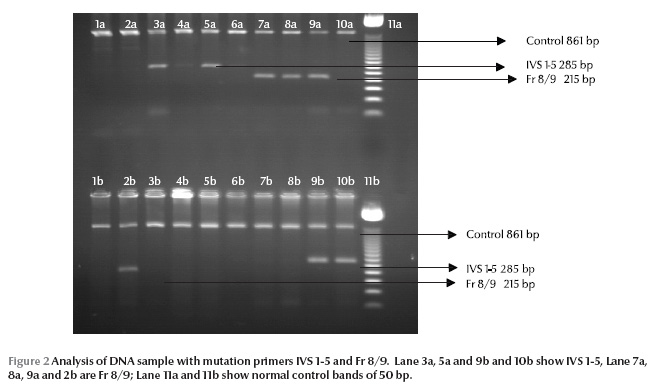
Other common mutations that were identified were CAP+1, IVS-II-1 (G–A), Cd 5 (–CT), Cd 15 (G–A), Cd 16 and Cd 30 and they were 12% of the total beta-thalassaemia genes (Table 2).
Discussion
Beta-thalassaemia is the most common genetic disorder in Pakistan. The carrier frequency of beta-thalassaemia is estimated to be around 6% in the Pakistani population [16]. Our results of molecular analysis of beta-thalassaemia syndrome in these 5 major ethnic groups revealed 5 common mutations which comprised 88% of the total beta-thalassaemia genes: IVS-1-5 (G–C), Fr 8/9 (+G), Fr 41/42 (–TTCT), IVS-1-1 (G–T) and Del 619. Other mutations identified during this study, which constituted 12% of the total beta-thalassaemia genes, were: CAP+1, IVS-II-1 (G–A), Cd 5 (–CT), Cd 15 (G–A), Cd 16 and Cd 30. Other molecular studies of beta-thalassaemia syndrome in Pakistan also showed the same 5 common mutations: IVS-1-5 (G–C), Fr 8/9 (+G), Fr 41/42 (–TTCT), IVS-1-1 (G–T) and Del 619 [13–16].
The spectrum of beta-thalassaemia mutations that was identified in the Indian population also showed the same 5 common mutations: IVS-1-5 (G–C), Fr 8/9 (+G), Fr 41/42 (–TTCT), IVS-1-1 (G–T) and Del 619. These accounted for 93.6% of the total beta-thalassaemia genes in the Indian population [19–23]. In the Pakistani Gujrati population (an Urdu-speaking population) the most common beta-thalassaemia gene was Del 619 which is also common in the Indian Gujrati population [20]. The similar pattern of molecular genetics of beta-thalassaemia syndromes in the Pakistani and Indian populations may be explained in 2 ways. First, Pakistan and India was one state for several hundred years before partition in August 1947 and cross-population and consanguineous marriages were common. The second reason is the large-scale migration from India to Pakistan and from Pakistan to India during partition.
There are also close similarities in the molecular genetics of beta-thalassaemia syndrome in the Arab and Pakistani populations. The molecular spectrum of beta-thalassaemia syndrome in United Arab Emirates (UAE) nationals revealed that IVS-1-5 (G–C) was the most common mutation, with a frequency of 66%. Other mutations that were reported in the UAE included: Fr 8/9 (+G), Cd 5 (–CT), IVS-II-1 (G–A), Cd 30 (G–C) and Cd 15 (G–A) [24,25]. The molecular spectrum of beta-thalassaemia in the Arab populations of Jordan, Egypt, Syrian Arab Republic, Lebanon, Yemen and Saudi Arabia revealed that the most frequent mutations were: IVS-1-5 (G–C), IVS-II-1 (G–A), IVS-1-1, Fr 8/9, Fr 41/42, Cd 15, Cd 16, Cap +1 (A–C), IVS-1-110, IVS-1-3′ end (-25 bp) and IVS-1-6 [25,26]. These similarities may be due to the influence of trading and population migration by the Arabs on the Pakistani populations of Sindh and Pakistani Punjab.
Conclusions
The beta-thalassaemia gene is present in all the major ethnic groups in the country (Punjabi, Pathan, Sindhi, Baluchi and Urdu speaking). Our molecular analysis of beta-thalassaemia syndrome in these 5 major ethnic groups has shown 5 common mutations which comprised 88% of the total beta-thalassaemia genes in the sample. These mutations included: IVS-1-5 (G–C), Fr 8/9 (+G), Fr 41/42 (–TTCT), IVS-1-1 (G–T) and Del 619. Other mutations we identified during this study constituted 12% of the total beta-thalassaemia genes and included CAP+1, IVS-II-1 (G–A), Cd 5 (–CT), Cd 15 (G–A), Cd 16 and Cd 30.
Beta-thalassaemia is the most common genetic disorder in Pakistan and this molecular analysis of beta-thalassaemia syndrome in Pakistan provides a baseline to help in the organization of a large-scale prevention programme based on prenatal diagnosis of beta-thalassaemia syndrome.
References
- Quek L, Thein SL. Molecular therapies in beta thalassaemia. British Journal of Haematology, 2007, 136(3):353–365.
- Pan HF et al. Current status of thalassemia in minority population in Guangxi, China. Clinical Genetics, 2007, 71(5):419–426.
- Bun HF et al. Hemoglobin: molecular genetics and clinical aspects. Philadelphia, WB Saunders, 1986.
- Cooley TB et al. A series of cases of splenomegaly in children with anemia and peculiar bones changes. Transactions of the American Pediatric Society, 1925, 37:29.
- Cooley TB et al. Erythroblastic anemia. American Journal of Diseases of Children, 1932, 43:705.
- Kulozik et al. The molecular basis of alpha thalassemia in India. Its interaction with the sickle cell gene. Blood, 1988, 71(2):467–472.
- Livingstone FB. Abnormal hemoglobin in human population. Chicago, Aldine, 1970.
- Verma IC et al. Multicenter study of the molecular basis of thalassemia intermedia in different ethnic populations. Hemoglobin, 2007, 31(4):439–452.
- Derakhshandeh-Peykar P et al. Distribution of beta-thalassemia mutations in Northern Province of Iran. Hemoglobin, 2007, 31(3):351–356.
- Aksoy M. Thalassemia intermedia; a genetic study in 11 patients. Journal of Medical Genetics, 1970, 7(1):47–51.
- Ibrahim SA et al. Thalassemia and high F-gene in Aleppo. Acta Haematologica, 1970, 44(5):287–291.
- Weatherall DJ. Molecular biology at the bedside. British Medical Journal, 1986, 292:1505–1508.
- Ahmed S et al. Molecular genetics of beta thalassaemia in Pakistan: a basis for prenatal diagnosis. British Journal of Haematology, 1996, 94(3):476–482.
- Baig SM et al. Prenatal diagnosis of beta-thalassaemia in Southern Punjab, Pakistan. Prenatal Diagnosis, 2006, 26(10):903–905.
- Ahmed S et al. Prenatal diagnosis of beta-thalassaemia in Pakistan: experience in a Muslim country. Prenatal Diagnosis, 2000, 20(5):378–383.
- Hafeez M et al. Regional and ethnic distribution of beta thalassemia mutations and effect of consanguinity in patients referred for prenatal diagnosis. Journal of the College of Physicians and Surgeons of Pakistan, 2007, 17(3):144–147.
- Newton CR et al. Analysis of any point mutation in DNA. The amplification refractory mutation system (ARMS). Nucleic Acids Research, 1989, 17(7):2503–2516.
- Old JM et al. Rapid detection and prenatal diagnosis of beta-thalassemia: studies in Indian and Cypriot populations in the UK. Lancet, 1990, 336:834–837.
- Varawalla NY et al. The spectrum of beta-thalassaemia mutations on the Indian subcontinent: the basis for prenatal diagnosis. British Journal of Haematology, 1991, 78(2):242–247.
- Vaz FE et al. Distribution of beta-thalassemia mutations in the Indian population referred to a diagnostic center. Hemoglobin, 2000, 24(3):181–194.
- Gupta A et al. Molecular genetic testing of beta-thalassemia patients of Indian origin and a novel 8-bp deletion mutation at codons 36/37/38/39. Genetic Testing, 2003, 7(2):163–168.
- Colah R et al. Impact of beta globin gene mutations on the clinical phenotype of beta thalassemia in India. Blood Cells, Molecules & Diseases, 2004, 33(2):153–157.
- Chakrabarti P et al. Spectrum of beta-thalassemia mutations in North Indian states: a beta-thalassemia trait with two mutations in cis. Clinical Biochemistry, 2005, 38(6):576–578.
- Quaife R et al. The spectrum of beta-thalassaemia mutations in the UAE national population. Journal of Medical Genetics, 1994, 31(1):59–61.
- El-Hazmi MA, Warsy AS, Al-Swailem AR. The frequency of 14 beta-thalassemia mutations in the Arab populations. Hemoglobin, 1995, 19(6):353–360.
- Baysal E. Molecular heterogeneity of beta-thalassemia in the United Arab Emirates. Community Genetics, 2005, 8(1):35–39.








